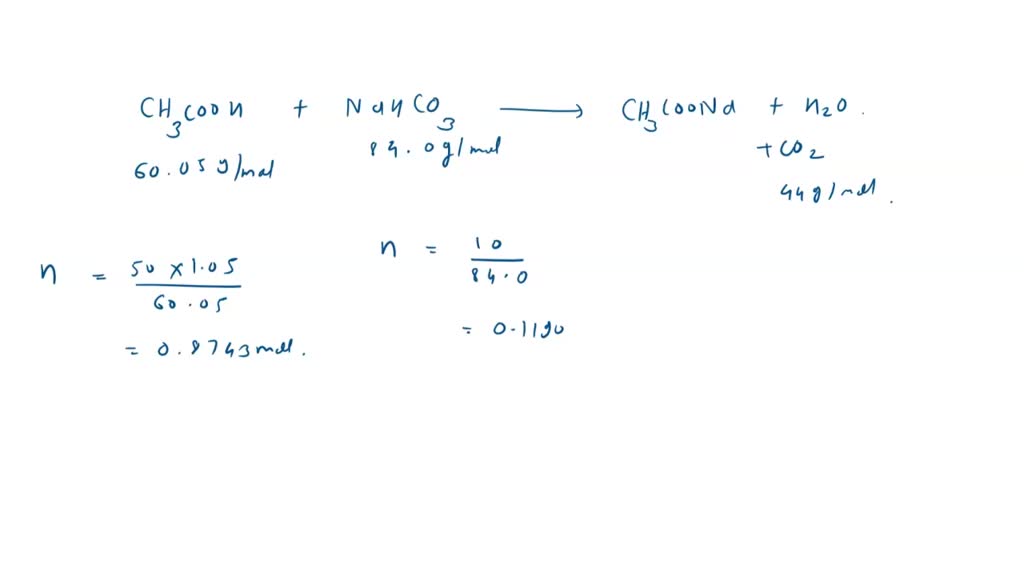
SOLVED: 10g of baking soda (Sodium Bicarbonate) is added to 50mL of vinegar (Density = 1.05g/mL). The resulting solution weighs 60.74g. How many liters of CO2 gas are released if it has

Multimedia: Controlling the Amount of Products in a Chemical Reaction | Chapter 6, Lesson 2 | Middle School Chemistry

SOLVED: 048 3.053 Datu: Mise baking soda (NaHCOz) used Mastt glove. baking soda vinegar REFORE MIXING Mass elove, baking soda vinegar AFTER MIXING Total volume of COz gas collected Nolumc afwatot bcakarl

Tap Density Tester,Kit To Measure The Baking Soda Density - Buy Tap Density Tester,Kit To Measure The Baking Soda Density,Density Tester Product on Alibaba.com

Dy-100b Tapped Bulk Density Analyzer,Tap Density Instrument,Kit To Measure The Baking Soda Density - Buy Bulk Density Analyzer,Tap Density Instrumen,Tapped Tester Product on Alibaba.com

Wuxi Beitang Chemical Light Raw Material Co., Ltd Sodium bicarbonate(baking soda)Wuxi Beitang Chemical Light Raw Material Co., Ltd

Water density science experiment || Water density changes when other substances dissolve in it - YouTube

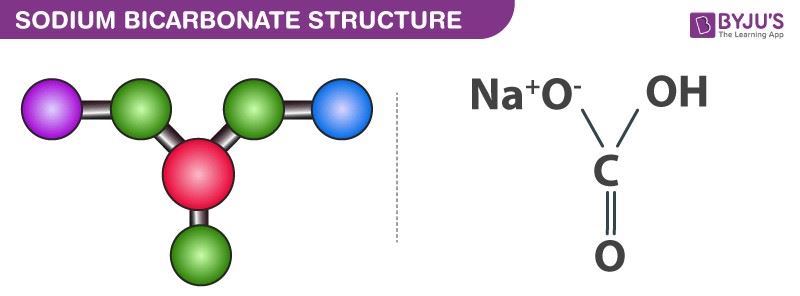
SOLVED:Baking soda (sodium bicarbonate, NaHCO3 ) reacts with acids in foods to form carbonic acid (H2 CO3), which in turn decomposes to water and carbon dioxide gas. In a cake batter, the




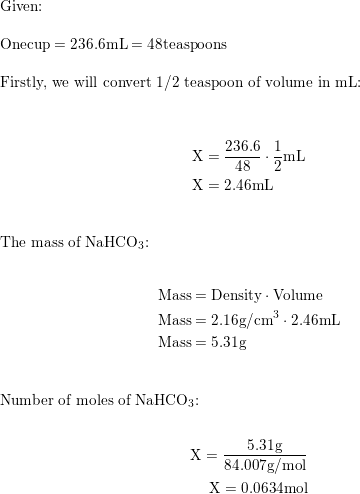







:max_bytes(150000):strip_icc()/baking-powder_annotated-5a72db68d14c4f03867d14ebf1f61055.jpg)


